UltraDrape® is a sterile, dual-action barrier and securement dressing for Ultrasound-Guided Peripheral Intravenous (UGPIV/USGPIV) procedures. It combines catheter securement and infection barrier protection in a single solution, improving patient safety and workflow efficiency for healthcare professionals.
UltraDrape® UGPIV Barrier and Securement
Product Subtitle
UGPIV Barrier and Securement
Key Features & Benefits
Made from high-quality, non-irritating materials and is not constructed with natural rubber latex, making it suitable for all patients and environments
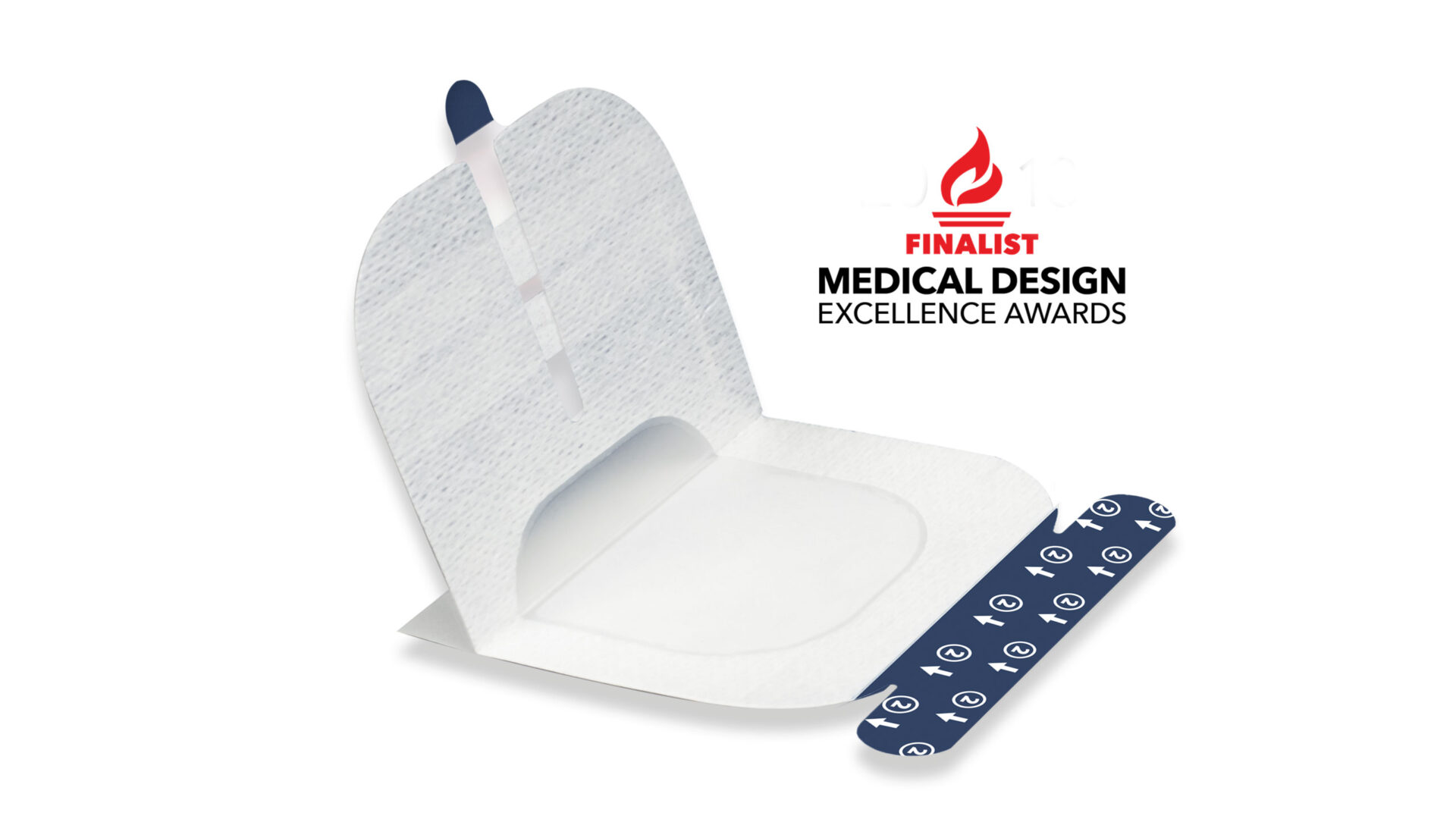
A single dressing serves as both a barrier to prevent contamination and a securement dressing for catheters, simplifying UGPIV procedures
The unique, “stand-alone” design prevents ultrasound gel from reaching the IV site while enabling seamless catheter insertion and securement
The protective film and adhesive strips enable a “no-touch” technique, are ASAP-approved to facilitate Standard-ANTT® (aseptic, non-touch technique), and help reduce infection risk while maintaining aseptic conditions
Reduces the need for separate sterile gels and covers, lowering procedure costs and cleanup time
The innovative design of UltraDrape™ ensures reliable catheter securement, reducing the likelihood of dislodgement and improving patient safety
By combining barrier and securement functions, UltraDrape™ streamlines the procedure and significantly reduces cleaning time after IV placement
Manufactured with a high-quality film layer, UltraDrape™ provides an effective barrier against all viruses and bacteria as small as 20nm, enhancing infection control
Each UltraDrape is sterile and individually packaged for convenience and safety
Videos
Available Configurations
Select a product number to view specific package details and specifications
Instructions for Use (IFU)
Download instructions for use files